Trusted Ketamine Purity Standards: Ultimate USP vs. EP vs. BP Guide
Authoritative Guide by Lyfeunit Technical SEO Team | Last Updated: April 2026
The Global Landscape of Ketamine Purity Standards
Pharmaceutical standards exist to ensure that regardless of where a drug is manufactured, its safety, potency, and quality remain consistent. For CAS 1867-66-9, these standards are codified in three major pharmacopeias: the United States Pharmacopeia (USP), the European Pharmacopoeia (EP), and the British Pharmacopoeia (BP).
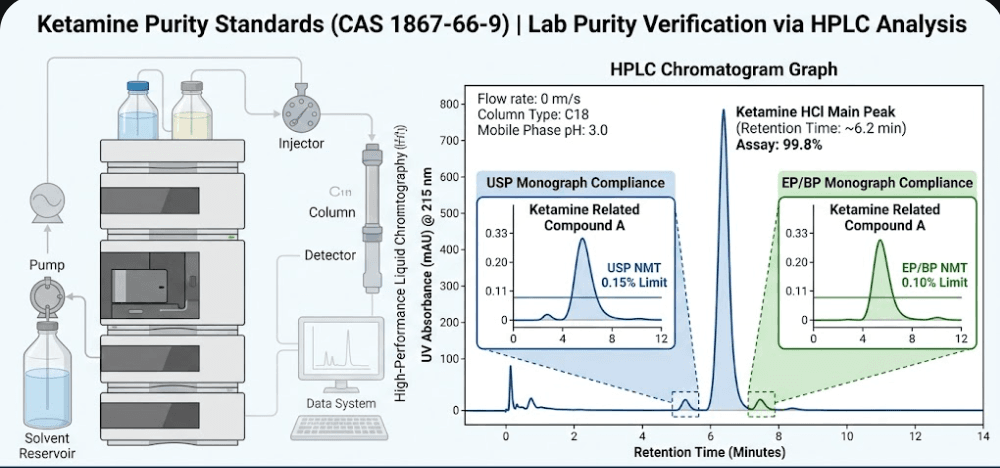
Figure 1: High-Performance Liquid Chromatography (HPLC) comparison showing different impurity detection thresholds under USP and EP Ketamine Purity Standards.
While these organizations strive for “harmonization,” significant nuances remain. These differences can impact everything from the Ketamine Hydrochloride impurity profile to the legal eligibility of a product in specific geographic markets. Understanding these details is essential for anyone seeking pharmaceutical grade ketamine for clinical or research purposes.
Comparison of Monograph Specifications
| Standard Parameter | USP Monograph | EP Monograph | BP Monograph |
|---|---|---|---|
| Assay (Dried Basis) | 98.5% – 101.5% | 99.0% – 101.0% | 99.0% – 101.0% |
| Individual Impurity | NMT 0.15% | NMT 0.10% | NMT 0.10% |
| Total Impurities | NMT 0.50% | NMT 0.30% | NMT 0.30% |
| Identification Method | IR & Chloride test | IR & Optical Rotation | IR & TLC |
1. Deep Dive into the USP (United States Pharmacopeia)
The USP is the authoritative body for pharmaceutical standards in the United States. Its monograph for Ketamine Hydrochloride focuses heavily on structural identity and “Related Compounds.” For a substance to be marketed as pharmaceutical grade ketamine in the US, it must pass the USP General Chapters <197K> (Infrared Absorption) and <191> (Physicochemical Tests for Identification).
Managing Ketamine Related Compound A
One of the primary markers monitored in the USP lab purity verification process is Ketamine Related Compound A. Chemically known as 1-[(2-chlorophenyl)(methylimino)methyl]cyclopentanol, this compound is an intermediate in the synthesis process. The USP sets a “Not More Than” (NMT) limit of 0.15% for this specific impurity. If a batch exceeds this, it cannot be classified as USP-grade, rendering it unusable for FDA-regulated medical applications.
2. The European Pharmacopoeia (EP) and Higher Stringency
In many technical circles, the EP standard is viewed as the “strict” sibling of the USP. The EP 11.0 standards for CAS 1867-66-9 require a tighter assay range (99.0% – 101.0%). Why the difference? The European Directorate for the Quality of Medicines & HealthCare (EDQM) prioritizes a lower threshold for unknown organic impurities to minimize potential adverse reactions in clinical settings.
3. The British Pharmacopoeia (BP): Localized UK Compliance
The BP remains a vital document for the UK market. While it heavily aligns with the EP, the BP includes specific appendices that address the formulation of Ketamine injections and oral solutions. For SEO strategists and lab procurement officers targeting the UK, identifying “BP Compliance” is a key signal of localized quality assurance.
4. The Importance of CAS 1867-66-9 in Analytical Chemistry
Using the Chemical Abstracts Service (CAS) registry number, CAS 1867-66-9, is the only way to avoid ambiguity in global trade. While “Ketamine” is a common name, the CAS number specifically identifies the Hydrochloride salt. During lab purity verification, the CAS number serves as the anchor for the Material Safety Data Sheet (MSDS) and the Certificate of Analysis (CoA).
5. Analyzing the Ketamine Hydrochloride Impurity Profile
The “Impurity Profile” is a comprehensive map of all substances within a sample that are not the active pharmaceutical ingredient (API). These can be categorized as:
- Organic Impurities: Synthesis intermediates like Ketamine Related Compound A or degradation products from improper storage.
- Inorganic Impurities: Residual reagents or heavy metals (lead, arsenic) often picked up during the manufacturing process.
- Residual Solvents: Volatile organic chemicals used in the crystallization process, such as toluene or hexane.
Modern lab purity verification uses HPLC with UV detection to ensure each of these categories falls below the pharmacopeial limits. For pharmaceutical grade ketamine, the total sum of these impurities must typically be less than 0.3% to 0.5%, depending on the monograph applied.
6. Best Practices for Lab Purity Verification
To achieve a high-ranking “Search Engine Optimized” and technically accurate result, labs must follow a rigorous 5-step workflow:
- Sample Preparation: Dissolving the API in a precise mobile phase (often a phosphate buffer).
- Chromatographic Separation: Using a C18 column to separate the molecules based on polarity.
- Detection: Monitoring the eluent at a specific wavelength (usually 215 nm).
- Quantitation: Comparing the peak areas of the sample against a certified Reference Standard.
- Documentation: Generating a CoA that explicitly states compliance with USP, EP, or BP.
Frequently Asked Questions (FAQ)
What defines “Pharmaceutical Grade” Ketamine?
Pharmaceutical grade ketamine is any batch that has been tested and certified to meet the minimum assay and maximum impurity thresholds defined by a recognized pharmacopeia (USP, EP, or BP). It must be produced in a cGMP (current Good Manufacturing Practice) facility.
If present in high concentrations, Ketamine Related Compound A can alter the safety profile of the drug. Its presence usually indicates an incomplete chemical reaction during synthesis or poor purification techniques.
Can a lab use both USP and EP standards simultaneously?
Yes. This is known as “Multi-Compendial” testing. Many global suppliers perform lab purity verification against both USP and EP standards to ensure their product can be sold in both the American and European markets without re-testing.
Why is the CAS 1867-66-9 number necessary for SEO?
From a digital marketing perspective, CAS 1867-66-9 is a “high-intent” search term. When a researcher searches for the CAS number rather than just the name, they are looking for technical data, making it a high-value keyword for conversion.
Conclusion: The Path to Technical Authority
Mastering the complexities of Ketamine Purity Standards is essential for maintaining safety and compliance in 2026. Whether you are conducting lab purity verification or sourcing pharmaceutical grade ketamine, understanding the nuances between USP, EP, and BP is non-negotiable. By adhering to the most stringent Ketamine Hydrochloride impurity profile limits, you ensure that your research or clinical applications are built on a foundation of absolute quality.
