ketamine Liquid
Pharmaceutical Grade Ketamine Liquid (CAS 1867-66-9)
Our Ketamine Liquid category provides high-purity aqueous solutions engineered for precise clinical research and pharmaceutical synthesis. Each formulation is prepared to USP Grade standards, utilizing the verified chemical entity CAS 1867-66-9 to ensure the highest levels of sterile integrity and molecular consistency for institutional applications.
Technical Specifications & Research Parameters
- Chemical Identity: Ketamine Hydrochloride (C13H17Cl2NO)
- Formulation Physicality: Clear, sterile aqueous solution optimized for stability and physiological pH balance.
- Quality Verification: Batch-certified purity confirmed through HPLC analytical protocols and rigorous pharmaceutical testing.
We prioritize data transparency and risk mitigation by providing detailed technical specifications and clinical safety signals. By focusing on the Science of Synthesis, we ensure that our liquid formulations meet the documentation requirements necessary for advanced studies into NMDA receptor antagonism and anesthetic research.
All liquid preparations are handled under strict pharmaceutical protocols to maintain the highest standards of excellence. Our commitment to regulatory compliance ensures that research institutions and practitioners receive a verified chemical product supported by comprehensive data and professional integrity.
Showing all 4 resultsSorted by popularity
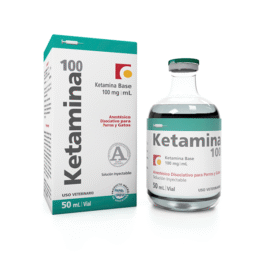
Ketamina 100mg/ml Vials | Veterinary Ketamine for Animals and Pets
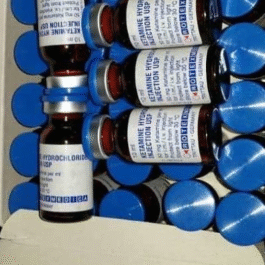
Price range: $650,00 through $13.500,00 Select options This product has multiple variants. The options may be chosen on the product pageRotexmedica Ketamine 500mg/10ml Vials | Mental Health Treatments
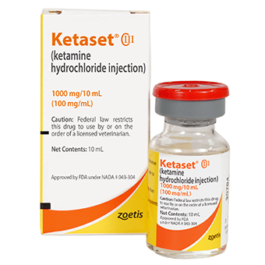
Price range: $675,00 through $14.000,00 Select options This product has multiple variants. The options may be chosen on the product pageKETASET Ketamine HCl Injection 1000mg/10ml
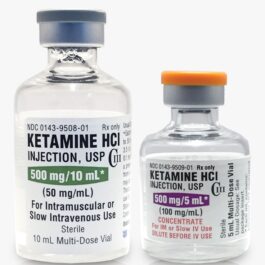
Price range: $675,00 through $13.000,00 Select options This product has multiple variants. The options may be chosen on the product pageBuy Ketamine HCL 500mg/10ml Vial USP Grade | LyfeUnit
Price range: $525,00 through $8.500,00 Select options This product has multiple variants. The options may be chosen on the product page
Recent Comments