Ketalar (Ketamine HCl) Injection 500mg/10mL: Comprehensive Technical Guide & Specifications
Ketalar (Ketamine HCl) Injection 500mg/10mL represents a critical component in modern anesthesia and emergency medicine. As a rapid-acting, non-barbiturate general anesthetic, it is uniquely positioned due to its profound analgesic properties and its ability to maintain stable hemodynamics. This USP-grade injection is formulated at a concentration of Ketamine 50mg/mL, providing a potent solution for general anesthesia induction and diagnostic sedation. By acting as a highly selective NMDA receptor antagonist, Ketalar produces a “dissociative” state that is distinct from traditional CNS depressants, making it an essential tool for licensed clinicians managing complex surgical and diagnostic cases.
Clinical Pharmacology: The NMDA Receptor Antagonist Mechanism
The therapeutic efficacy of Ketalar (Ketamine HCl) Injection 500mg/10mL is rooted in its complex interaction with the central nervous system. Unlike volatile anesthetics or GABAergic agents like propofol, Ketamine Hydrochloride USP primarily targets the N-methyl-D-aspartate (NMDA) receptor complex. As an NMDA receptor antagonist, it binds to the phencyclidine site within the ion channel, effectively blocking the excitatory effects of glutamate. This action occurs primarily in the thalamocortical and limbic systems, leading to what is clinically defined as a dissociative anesthetic state.
During this state, the patient’s association pathways are interrupted, preventing the brain from processing sensory input into conscious perception. This results in significant analgesia while maintaining protective airway reflexes, such as coughing and swallowing. Furthermore, Ketalar provides “technical proof” of its clinical superiority in specific scenarios through its sympathomimetic activity. It stimulates the cardiovascular system, leading to a modest increase in heart rate and blood pressure, which is particularly advantageous in patients where hypotension must be avoided.
Product Composition and Pharmaceutical Standards
To ensure pharmaceutical-grade precision, each multi-dose vial 500mg of Ketalar is manufactured under rigorous quality control standards. This USP grade injection is a sterile, colorless solution that is slightly acidic, with a pH range of 3.5 to 5.5. The formulation is designed for high stability and reliability in parenteral administration.
- Active Ingredient: Ketamine Hydrochloride USP equivalent to 50mg Ketamine base per mL.
- Concentration: 500mg per 10mL (50mg/mL).
- Excipients: Includes 0.1 mg/mL Benzethonium Chloride as a preservative to ensure the integrity of the multi-dose container.
- Chemical Structure: (±)-2-(o-chlorophenyl)-2-(methylamino) cyclohexanone hydrochloride.
- Regulatory Status: Strictly classified as a CIII controlled substance and a DEA Schedule III Anesthetic.
Indications for Use: Beyond General Anesthesia Induction
Ketalar (Ketamine HCl) Injection 500mg/10mL is a versatile diagnostic sedation agent with a wide range of indications. Its primary role is as the sole anesthetic agent for surgical and diagnostic procedures that do not require skeletal muscle relaxation. Because of its rapid onset and high potency, it is often the first choice for:
- General Anesthesia Induction: Providing a fast transition to unconsciousness before the administration of secondary maintenance agents.
- Supplementation: Enhancing the effects of low-potency anesthetics like nitrous oxide.
- Emergency Procedures: Ideal for rapid sequence induction (RSI) in trauma settings where cardiovascular stability is paramount.
- Pediatric Anesthesia: Widely utilized in pediatric settings for minor surgical procedures due to its safety profile regarding respiratory drive.
Detailed Dosage and Administration Protocols
The administration of parenteral ketamine requires meticulous titration based on the patient’s weight, age, and clinical status. For general anesthesia induction via the intravenous (IV) route, the initial dose typically ranges from 1 mg/kg to 4.5 mg/kg. An intravenous dose of 2.0 mg/kg is usually sufficient to produce 5 to 10 minutes of surgical anesthesia within 30 seconds of injection.
For intramuscular (IM) administration, the dose is higher, ranging from 6.5 mg/kg to 13 mg/kg. The 500mg/10mL concentration allows clinicians to deliver these doses efficiently. It is imperative that IV administration occurs at a slow rate (not exceeding 0.5 mg/kg/min) to minimize the risk of transient respiratory depression or a sharp increase in blood pressure. Maintenance of anesthesia is achieved by repeated increments of half to the full induction dose, depending on the duration of the procedure.
Pharmacokinetics: Absorption, Distribution, and Metabolism
Understanding the pharmacokinetics of Ketalar (Ketamine HCl) Injection 500mg/10mL is essential for effective clinical management. Following administration, Ketamine is rapidly distributed into highly vascularized tissues, such as the heart and brain, followed by redistribution into less vascularized muscle and adipose tissue. This rapid redistribution is what accounts for the relatively short duration of the anesthetic effect.
The metabolism of Ketamine Hydrochloride USP occurs primarily in the liver through a process called N-demethylation. The primary metabolite, norketamine, possesses approximately 33% of the anesthetic potency of the parent compound. This metabolite is later hydroxylated and conjugated before being excreted via the kidneys. This metabolic pathway is a key “technical proof” point, as clinicians must account for potential prolonged effects in patients with impaired hepatic or renal function.
Safety Profile: Adverse Reactions and Contraindications
While Ketalar (Ketamine HCl) Injection 500mg/10mL is highly effective, it is associated with a distinct set of adverse reactions. The most notable is “emergence delirium,” which occurs as the patient recovers from the dissociative anesthetic state. This may include vivid dreams, confusion, or hallucinations. These reactions can be minimized by maintaining a quiet recovery environment or pre-administering benzodiazepines.
Cardiovascular effects, such as transient increases in blood pressure and pulse rate, are common and usually self-limiting. However, Ketamine is contraindicated in individuals for whom a significant rise in blood pressure would constitute a serious hazard, such as those with severe cardiovascular disease or uncontrolled hypertension. Additionally, clinicians should monitor for increased intracranial pressure (ICP) and intraocular pressure in susceptible patients.
Drug Interactions and Chemical Compatibility
Ketalar (Ketamine HCl) Injection 500mg/10mL exhibits specific interactions with other clinical agents. Prolonged recovery times may be seen when used in conjunction with barbiturates or narcotics. It is chemically incompatible with barbiturates and diazepam; therefore, these agents should not be mixed in the same syringe or infusion line, as precipitation will occur.
Frequently Asked Questions (FAQ)
What is the exact concentration of Ketalar (Ketamine HCl) Injection 500mg/10mL?
The Ketalar (Ketamine HCl) Injection 500mg/10mL vial is formulated at a concentration of 50mg/mL. This high-potency parenteral solution is designed for both intravenous (IV) and intramuscular (IM) administration, providing clinicians with a versatile tool for rapid general anesthesia induction and procedural sedation.
Why is Ketalar classified as a dissociative anesthetic?
Ketalar is defined as a dissociative anesthetic because it interrupts association pathways in the brain rather than causing generalized CNS depression. This unique state is achieved through its role as an NMDA receptor antagonist, allowing patients to remain physically stable with intact airway reflexes while being insensitive to pain and unaware of their surroundings.
What is the regulatory status of Ketamine Hydrochloride USP?
In the United States, Ketamine Hydrochloride USP is strictly regulated as a CIII controlled substance. This DEA Schedule III Anesthetic designation requires precise record-keeping, secure storage in a locked cabinet, and administration only by licensed healthcare professionals in a controlled clinical environment.
How does the NMDA receptor antagonist mechanism provide “technical proof” of efficacy?
The NMDA receptor antagonist mechanism is the cornerstone of Ketamine’s clinical profile. By binding to the NMDA receptor’s phencyclidine site, Ketalar inhibits glutamate-mediated excitatory signaling. This specific action provides technical proof of its ability to induce rapid analgesia and anesthesia while maintaining cardiovascular stimulation, which distinguishes it from other anesthetic agents.
What are the primary indications for using parenteral ketamine in surgery?
Parenteral ketamine is primarily indicated as the sole anesthetic agent for short diagnostic and surgical procedures. It is also an essential agent for general anesthesia induction prior to using other agents, and as a supplement to low-potency anesthetics like nitrous oxide, especially when cardiovascular stability is a priority.
How should Ketalar (Ketamine HCl) Injection 500mg/10mL be stored?
To maintain the stability of the USP-grade injection, vials must be stored at controlled room temperature, specifically between 20° to 25°C (68° to 77°F). It is vital to protect the solution from light and avoid exposure to extreme heat to ensure the chemical integrity of the 500mg multi-dose vial.
Regulatory Compliance and Storage Standards
As a CIII controlled substance, the procurement and storage of Ketalar (Ketamine HCl) Injection 500mg/10mL are strictly regulated by the DEA. It must be stored at controlled room temperature between 20° to 25°C (68° to 77°F). Vials must be protected from light and kept in a secure, locked cabinet to prevent unauthorized access. Lyfeunit ensures that every DEA Schedule III Anesthetic provided meets these rigorous compliance benchmarks to maintain the highest levels of professional and technical authority.
Medical Disclaimer
The information contained herein regarding Ketalar (Ketamine HCl) Injection 500mg/10mL is intended solely for licensed medical professionals and pharmaceutical practitioners. This technical description serves as an authoritative guide for Ketamine Hydrochloride USP and is not a substitute for professional clinical training.
This product is a CIII controlled substance and must be administered only by personnel experienced in the induction of general anesthesia, airway management, and the use of resuscitative equipment. Lyfeunit does not provide medical advice or endorse self-administration. Improper use of a dissociative anesthetic like Ketamine can lead to severe adverse events, including respiratory depression, cardiovascular stress, and psychological distress. Always refer to the full prescribing information and local regulatory guidelines before clinical use.
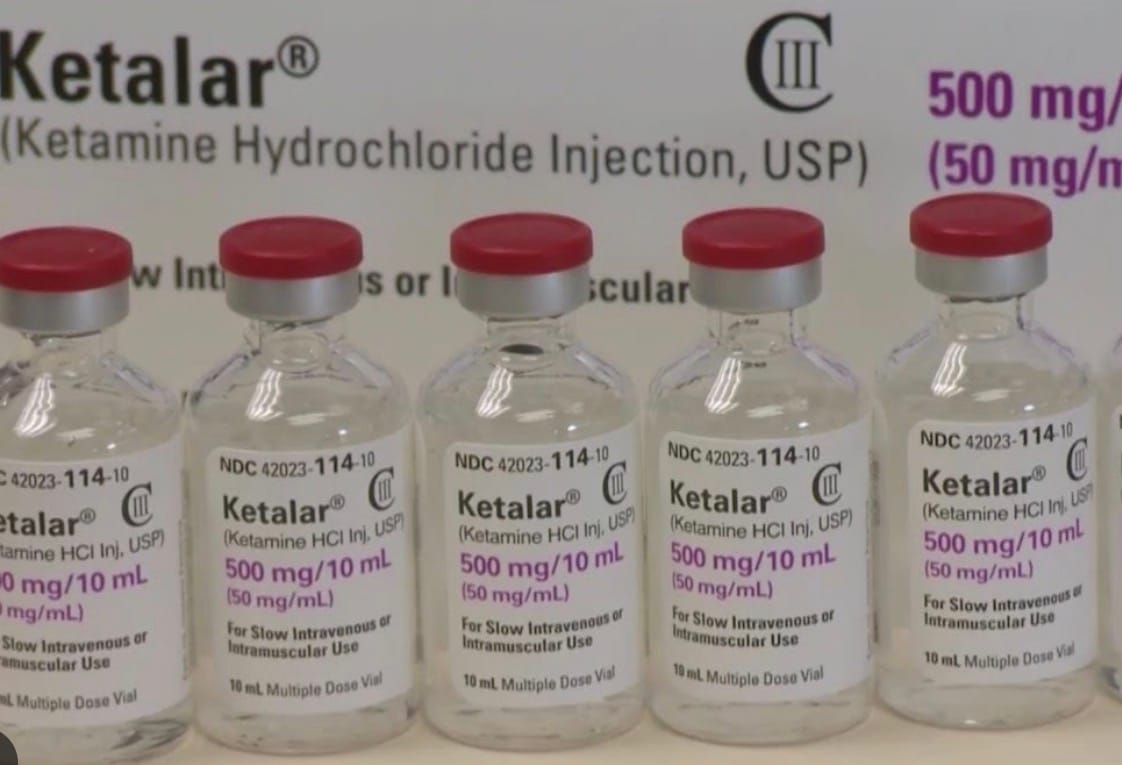

Dependable Sympathomimetic Profile for Emergency Induction –
As a practicing anesthesiologist in an urban trauma center, Ketalar (Ketamine HCl) remains an indispensable tool in our rapid-sequence induction protocols. What makes the 500mg/10ml concentration so vital in emergency settings is its ability to maintain hemodynamic stability. Unlike other induction agents that can cause a severe drop in blood pressure, Ketamine’s transient cardiovascular stimulation is highly beneficial for hypovolemic patients. The USP standard guarantees the strict purity and consistency we require when every second counts.
A Life-Changing Clinical Protocol for Chronic Pain Management –
After years of managing severe CRPS (Complex Regional Pain Syndrome), my pain specialist initiated a high-dose clinical Ketamine infusion protocol using USP-grade Ketalar. Sitting in a monitored medical environment while receiving this medication intravenously was intense, but the NMDA-receptor blockade effectively ‘reset’ my pain pathways. For anyone considering this, make sure you go through a fully licensed clinic—the formulation matters immensely, and having a professional team manage the dosage is everything.
Standardizing Consistency in Sub-Anesthetic Infusion Therapy –
Managing a clinical wellness space that offers off-label ketamine infusions for treatment-resistant depression requires meticulous quality control. We strictly utilize USP-grade Ketamine HCl 50mg/mL formulations to calculate precise, weight-based sub-anesthetic doses (typically 0.5 mg/kg over 40 minutes). Having a reliable, standardized product ensures that our infusion pumps deliver uniform results, mitigating adverse psychotomimetic spikes and keeping patient safety at the forefront of our practice.